Dream Team Discovers Potential New Weapon Against Neuroblastoma
Formerly known as the St. Baldrick’s – Stand Up to Cancer Pediatric Cancer Dream Team, this team is now the St. Baldrick’s EPICC Team (Empowering Pediatric Immunotherapies for Childhood Cancer).
Last week, in the pages of the medical journal Cancer Cell, St. Baldrick’s researchers announced a discovery that could radically transform treatment for kids with neuroblastoma – a new immunotherapy drug candidate that harnesses the immune system to fight cancer.

Neuroblastoma is a cancer that begins in the nerve tissue outside the brain, usually in a child’s abdomen. It strikes very young children, up to about age 7, and is the most common cancer diagnosed in infants. Only about 50% of patients survive the high-risk form of neuroblastoma.
All of this makes this new targeted immunotherapy for neuroblastoma especially good news, but it gets even better.
Immunotherapy Q&A with St. Baldrick’s Researcher, Dr. Grupp
We sat down virtually with St. Baldrick’s researcher, Stephan A. Grupp, MD, PhD, for a Q&A on immunotherapy. Dr. Grupp, located at Children’s Hospital of Philadelphia, is a key member of the Stand Up to Cancer – St. Baldrick’s Pediatric Dream Team*. He treated the first child with CAR T cell therapy, which was recently approved by the FDA for acute lymphoblastic leukemia (ALL). Check out our Twitter chat with Dr. Grupp and see what he thinks the future is for immunotherapy and childhood cancers.
What Is Immunotherapy?
St. Baldrick’s Scholar Dr. David Barrett gives us an inside look at immunotherapy, and we meet the first pediatric patient to ever receive this new cancer treatment.

Emily Whitehead, the first pediatric patient to be treated with immunotherapy, 18 months post-treatment and starting third grade.
For most of us, our immune systems fight off disease every day. A healthy immune system can even fight off life-threatening diseases like cancer. And when we do get sick, a healthy immune system attacks sickness and help us get back to feeling like ourselves again.
It is a different story for kids with cancer. A child with cancer has a suppressed immune system. It’s as if a blindfold has been pulled down, and the body is unable to target invasive cells. You might say the immune system is asleep.
What if doctors could wake up a sleeping immune system?
BIG NEWS: FDA Approves Groundbreaking CAR T Cell Therapy for Kids With Leukemia
Formerly known as the St. Baldrick’s – Stand Up to Cancer Pediatric Cancer Dream Team, this team is now the St. Baldrick’s EPICC Team (Empowering Pediatric Immunotherapies for Childhood Cancer).
St. Baldrick’s supporters, this is a day to celebrate! You have helped make history. Today, the FDA approved the first gene therapy available in the United States. Called Kymriah, it’s an entirely new way of treating cancer and it’s saving lives.

This “living drug” is for patients with a type of acute lymphoblastic leukemia (ALL), a cancer of the blood and bone marrow and the most common form of childhood cancer. Scientists genetically modify a patient’s own immune cells in the lab, then infuse these new cells back into the patient’s body. These modified cells – called CAR T cells or chimeric antigen receptor T cells – then prompt the child’s own immune system to attack and kill leukemia cells.
CAR T Cell Therapy and a Turning Point in Treatment: News From the Dream Team
Formerly known as the St. Baldrick’s – Stand Up to Cancer Pediatric Cancer Dream Team, this team is now the St. Baldrick’s EPICC Team (Empowering Pediatric Immunotherapies for Childhood Cancer).
As one of the most exciting projects supported by St. Baldrick’s – the SU2C-St. Baldrick’s Pediatric Cancer Dream Team – nears the end of its funding, a group of expert reviewers met at Stanford University on July 14th to evaluate its impact to date. To say they were impressed would be an understatement.

Members of the Dream Team gather at a meeting in July to discuss the impact of their research efforts.
The day began with a report from Dr. Stephan Grupp, director of the Cancer Immunotherapy Program at Children’s Hospital of Philadelphia.
He had flown to the Dream Team meeting straight from the Maryland headquarters of the Food and Drug Administration (FDA), where hundreds had packed a hearing room the day before. An FDA advisory panel was considering what could soon be the first gene therapy to be marketed in the United States, pioneered by Dr. Grupp and his colleagues.
Stop, Collaborate and Listen: How Teamwork Makes the Dream Team Work
Formerly known as the St. Baldrick’s – Stand Up to Cancer Pediatric Cancer Dream Team, this team is now the St. Baldrick’s EPICC Team (Empowering Pediatric Immunotherapies for Childhood Cancer).
When people come together for a cause, incredible things happen. That’s true for St. Baldrick’s head-shaving events AND for the lifesaving work done by St. Baldrick’s researchers, especially the Stand Up To Cancer – St. Baldrick’s Pediatric Dream Team. As they say, teamwork makes the dream work! Read on to learn more about how cooperation and sharing between these researchers means big advances for kids with cancer.

They say two heads are better than one. But what about 149? That is how many brilliant brains are working together to conquer childhood cancers as part of the SU2C – St. Baldrick’s Pediatric Cancer Dream Team.
And it’s that cooperation that’s accelerating progress for kids and laying the foundation for better treatments and for cures, said Dr. Crystal Mackall and Dr. John Maris, co-leaders of the Dream Team.
The Kid With Superhero Cells: Leon’s Immunotherapy Story
Honored Kid Leon is one tough cookie. He’s fought childhood cancer not just once, but twice, and this time, Leon and his family hope it’s gone for good — thanks to an immunotherapy trial run by Leon’s buddy Dr. Daniel Lee, an investigator with the Stand Up to Cancer – St. Baldrick’s Dream Team*.

Leon and St. Baldrick’s researcher Dr. Daniel Lee share some smiles.
Everything changed one hot summer day in Colorado. Leon was spraying his cousins with a water gun and playing with the hose. Then the 9-year-old slipped on the slick deck and fell hard.
It was a badly bruised hip, said the doctors in the emergency room. It will heal. But it didn’t. Leon’s grandmother, Lisa, watched her normally active grandson walk gingerly and even resort to crutches.
Then she watched Leon get tired more quickly than a kid should. And then she watched him sleep. He slept and slept.
“And I knew then,” Lisa said. “I was like, ‘I think he’s sick again.’”
‘Pura Vida!’: Phineas’ Summer in Costa Rica
Ambassador Phineas’ dad, Carlos, shares what the family has been up to this past summer, and he looks back at where their family was 10 years ago — and where they might be now if it weren’t for the immunotherapy clinical trial that saved Phineas’ life.

Ten years ago I was nearing the end of the worst summer of my life.
‘My One Ticket to Live On’: How a Clinical Trial Saved Mitch Carbon’s Life
Like most college freshmen, Mitch Carbon is excited to be getting a fresh start. But unlike his peers, just two years ago, he didn’t think he’d live to see this day. Read on for more of Mitch’s story and the clinical trial that saved his life — all made possible by YOU.
When Mitch Carbon was a junior in high school, he was preparing to die.
The History of Childhood Cancer Research
In honor of Childhood Cancer Awareness Month, we are looking back through the decades at the milestones in research that have brought us to where we are today, and we are looking to the future — made brighter for kids with cancer because of you!
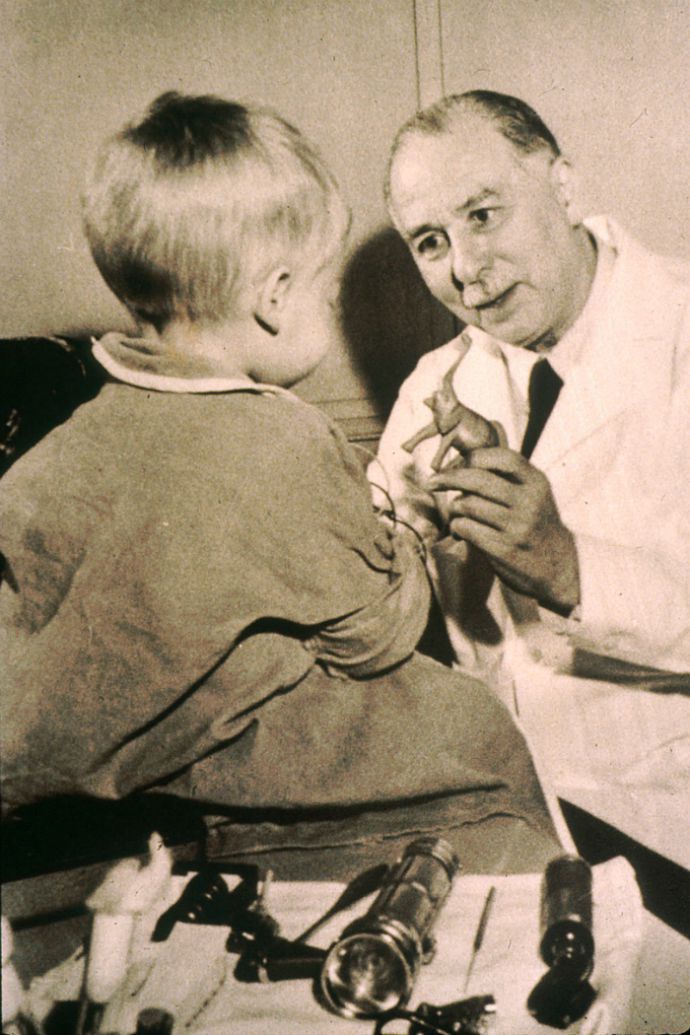
Dr. Sidney Farber examines a young patient. Photo from the National Institutes of Health
The childhood cancer research world we know today owes itself to the progress of the 1950s and 1960s.
It was a time “when people really began to believe that you could take a swipe at cancer,” said Dr. Crystal Mackall of the National Cancer Institute. Dr. Mackall is co-chair of the Stand Up to Cancer – St. Baldrick’s Pediatric Cancer Dream Team*, which is working to develop new, targeted therapies for kids with difficult-to-treat cancers.
« Newer PostsOlder Posts »

 SBF
Tweets »
SBF
Tweets »