

Dr. Parsons, a genomics expert on the Stand Up To Cancer – St. Baldrick’s Pediatric Cancer Dream Team*.
For example, we know that childhood cancer is caused by genetic mutations. What we don’t know is how or why most of those mutations occur.
And we’re still trying to figure out what the mutations mean — in terms of the cancer and its ability to thrive, and in terms of our bodies and their ability to overcome disease.
That’s the focus of genomics, explains Dr. Donald Parsons, the principal investigator at Baylor College of Medicine for the Stand Up To Cancer – St. Baldrick’s Pediatric Cancer Dream Team*.
“Genomics is the study on a large scale of all the genetic changes that occur in a patient’s DNA,” Dr. Parsons says. “It really tries to look at — in a single patient or across a group of patients — all the different changes that occur and how they might interact with each other.”
Genes, Proteins, and Mutations
Genomics research often goes like this: A researcher looks for a mutation in the DNA, and once they find one, they try to find out everything they can about the gene it’s in, Dr. Parsons explains.
Our bodies contain about 20,000 genes. Each gene carries the blueprints that make one or more specific proteins. Another way to think about this is that the proteins carry out the function of the genes. Everything you do — laughing, breathing, digesting your food — happens because of genes and their proteins.
Some mutations can cause a gene to make a defective protein. Some can cause it to make too much or too little of a protein. Some can stop it from making a protein altogether.

Rows of chromosomes at the Science Museum in London. Photo by George Gastin.
“We also need to then figure out how many patients have that mutation and if there’s some link between that and how patients respond to specific treatments,” he adds.
For example, scientists have identified a mutation that is present in over 80% of alveolar rhabdomyosarcoma (aRMS) cases. They believe that mutation is responsible for telling the cancer to grow and spread. We know that this mutation isn’t the sole cause of aRMS — otherwise, it would be present in 100% of the cases — but it’s a very important factor, and patients with this mutation are less likely to respond to traditional therapies. One St. Baldrick’s researcher is using this information to develop a drug to directly target the mutation, which will hopefully yield a more effective treatment for this aggressive form of childhood cancer.
But even without drugs to target the mutations, just knowing that the mutations are associated with poorer outcomes in the clinic can help doctors decide to put these patients on a more rigorous treatment plan from the outset, increasing their chance of survival in the long run.
Cracking the Code
As promising as it sounds, genomics research is not without its challenges.
Genomics is a relatively young field of research. It can be traced back to Fred Sanger, aptly dubbed “the father of genomics,” whose novel DNA sequencing technique earned him the 1980 Nobel Prize in Chemistry. He was also the first to decode the entire genome, or all the DNA, of a living organism — phi X 174, a virus that grows in bacteria.
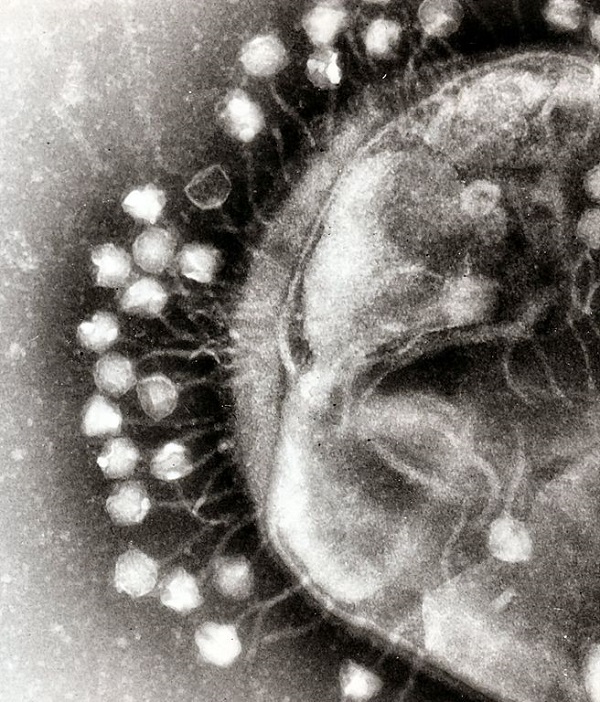
An electron micrograph of viruses attached to a bacterial cell wall.
Unfortunately, many of its pages remain gibberish. We know what all the genes are supposed to look like, but we don’t always know how they work or what they do.
Finding the meaning in all the pages of the manual is the next task, and it’s a big one.
One of the challenges in using genomics to study childhood cancer is that we don’t know what we don’t know. A researcher might find only two genetic mutations in the tumor she is studying, but how does she know when to stop looking for more?
“We are still learning about what tests are necessary to understand the full spectrum of mutation types in each type of cancer,” Dr. Parsons explains.
The Challenge of Small Numbers
There are multiple types of pediatric cancers, and many of them have several subtypes, each with their own biology and unique sets of mutations. “There are some types of cancer where you look for all the mutations in their genome and you see a relatively large number, and there are others where you see almost none,” Dr. Parsons says. “Pediatric cancers are more often the latter type, and what we are trying to understand is whether we already know everything we need to know about the genetics of some of those tumors — that they’re just pretty simple in terms of their biology and so it’s OK that there are not many mutations — or it may be that for some of those tumors, we just haven’t looked in the right places yet.”
That’s one reason why genomics researchers want to compare the genomes for different patients. But that leads to another big challenge: what Dr. Parsons calls “the challenge of small numbers.”
That’s the idea behind the Dream Team*: if we can pool all our data, theories, evidence, and discoveries together, then we’re bound to get better results — and faster.
“In terms of pediatric cancers, you’re talking about tens or hundreds of patients a year [for each type or subtype of cancer],” Dr. Parsons says. “And so getting the samples that you need for study and finding researchers to collaborate with to obtain those samples can be a real roadblock.”
Speeding Progress With Teamwork
But that’s why researchers in his field are doing a lot of collaborative work, he explains. And that’s the idea behind the Dream Team*: if we can pool all our data, theories, evidence, and discoveries together, then we’re bound to get better results — and faster.
Genomics plays a crucial role in the work of the Dream Team*. Dream Team* researchers have already developed a novel immunotherapy treatment approach that harnesses the body’s own immune system to attack cancer cells. But in order to develop those therapies, researchers first need to identify a target that’s present on cancer cells and not on healthy cells.
That’s where genomics comes in.
Once these new therapies enter the clinic, researchers are using genomics to try to understand why they work for some patients and not for others.
Dr. Parsons sees genomics as the key to understanding and improving treatment outcomes for some of the most deadly pediatric cancers.
“When we give these immunotherapies to patients, there are some who respond much better than others, and we often don’t really have a good explanation for that,” Dr. Parsons says. “So the other job for genomics in this project is to try and figure out why some patients’ tumors or immune systems are different than others.”
Dr. Parsons sees genomics as the key to understanding and improving treatment outcomes for some of the most deadly pediatric cancers.
“There are a lot of tumors and cancers that we’ve gotten really good at treating, although the treatments we give are more harsh for patients than we’d like. But there’s a subset of tumors that we just haven’t gotten much better at curing — some of the brain tumors and some of the other metastatic tumors, for example,” he explains. “So our other goal and part of the real rationale behind the Dream Team* is to try to develop therapies specifically for these very hard-to-treat childhood cancers.”
Immunotherapy has always sounded like an excellent approach in theory, according to Dr. Parsons. But now, with success stories like Phineas’, we’re seeing proof that it can actually put patients into remission that couldn’t have gotten there otherwise.
“It’s a really exciting time,” Dr. Parsons says. “We’ve started to see that it can work, and now we just have to figure out the best ways to make it work.”
The answer, it seems, lies in our genes.
Help fund the research that can lead to breakthroughs and cures for kids with cancer. Donate today.
*Formerly known as the St. Baldrick’s – Stand Up to Cancer Pediatric Cancer Dream Team, this team is now the St. Baldrick’s EPICC Team (Empowering Pediatric Immunotherapies for Childhood Cancer).
Read more stories on the St. Baldrick’s blog:

 SBF
Tweets »
SBF
Tweets »